|
Such structural formulae which focus only on the valence electrons involved in bond formation are called complete structural formulae.Įntrancei Chemistry Doubts page consist of more questions for reference. oxygen, nitrogen, sulphur, phosphorus, halogens) may or may not be shown. Lone pair of electrons on hetero atoms (e.g. 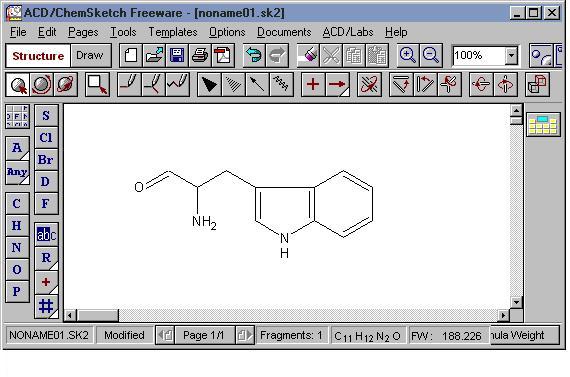 
Non bonding electrons, like bonding electrons, almost always come in pairs called lone pairs. These are assigned to specific atoms and are represented by dots drawn next to the symbols for these atoms. The valence electrons that are not included in covalent bonds are called nonbonding electrons. A triple bond is represented by three dashes between the atoms. A double covalent bond, in which two pairs of electrons are shared, is shown by two dashes between the atoms. When there is one dash between two atoms, the atoms are said to be bonded by a single covalent bond. The Lewis structure can be simplified by representing a shared electron pair by a ‘stick’ (dash, -) between the bonded atoms. 
Molecule editors can manipulate chemical structure representations in either a simulated two-dimensional space or three-dimensional space, via 2D computer graphics or 3D computer graphics, respectively. What is a Dash structural formula give an example? Chemistry Doubts A molecule editor is a computer program for creating and modifying representations of chemical structures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
